At this point, we are all familiar with SARS-CoV-2, the virus that causes COVID-19, and its infamous spike protein, which it uses to crack open our cells and crank out copies of itself like a horrifying Xerox machine.
The spike has been a big target of scientific interest: two vaccines, by Pfizer and Moderna, are based on it and have recently reported efficacies of over 90%.
But for treating COVID-19, there are other proteins inside our tiny foe that may be better targets.
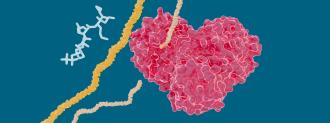
One is an enzyme called the main protease.
When SARS-CoV-2 is hijacking your cells to replicate itself, it creates long chains, called polyproteins. But in order for them to do anything, these chains need chopped up into individual workable proteins — that’s the main protease’s job.
Stop the main protease from slicing the proteins, stop the virus’s replication.
Now, researchers at the Oak Ridge National Laboratory (ORNL) believe that hepatitis C drugs may be able to do just that.
Looking to the Cures We Already Have
Their study, published in Structure, used room temperature x-rays to create a detailed, 3D model of the main protease, showing the exact arrangement of its atoms and where the hep C drug’s chemical bonds would occur.
“What we found is that hepatitis C drugs bind to and inhibit the coronavirus protease,” lead author Daniel Kneller said in a press release. “This is an important first step in determining whether these drugs should be considered as potential repurposing candidates to treat COVID-19.”
The team studied three protease inhibitors already approved by the FDA for treating hepatitis C — boceprevir, narlaprevir, and telaprevir.
Debuting in 2011, a combination of these drugs can now cure about 95% of hepatitis C infections — an amazing achievement for a deadly disease that was only discovered in 1989.
Hepatitis C is the first (and so far only) chronic viral disease to be cured — and protease inhibitors were the key.
To see how effective these drugs were at binding to the main protease, they used a process called “in vitro enzyme kinetics” to study how well the drugs bind to SARS-CoV-2 in a test tube. Drugs with a high “binding affinity” stick better to the main protease.
They found that the hepatitis C drugs (especially boceprevir and narlaprevir) had a high binding affinity — and therefore, the potential to gum up that main protease.
Repurposing Drugs
Drugs we already know well and have already been studied and approved by the FDA are our best shot of rapidly finding a good COVID-19 treatment.
“What we’re doing is laying the molecular foundation for these potential drug repurposing inhibitors by revealing their mode of action,” said ORNL’s Andrey Kovalevsky.
“We show on a molecular level how they bind, where they bind, and what they’re doing to the enzyme shape. And, with in vitro kinetics, we also know how well they bind. Each piece of information gets us one step closer to realizing how to stop the virus.”
They also found out something unusual about the main protease in the process: it can change its shape, altering itself with the size and structure of the inhibitor drugs glommed on to it.
As per usual, the COVID caveats apply. This is early work; it has identified potential drug candidates, not a proven cure.
Another wrinkle: hep C drugs are wildly expensive, which may make them less-than-ideal options in a practical sense.