MIT researchers have turned a system found in bacteria into programmable “nanosyringes” for injecting proteins into individual human cells — potentially overcoming a huge problem in medicine.
The challenge: We have around 20,000 different types of proteins performing countless functions in our bodies — these molecules construct our cells, regulate our genes, power our immune systems, transport oxygen in our blood, and countless other functions.
Genetic mutations or other damage can mess up our proteins, though — too much, too little, or the wrong shape can cause all kinds of health problems. On the flip side, if we could provide the right protein, in the right cells, we could treat or cure many diseases, too.
While every cell in our bodies naturally makes proteins, getting proteins into cells from the outside is incredibly difficult, because these molecules are so big — this makes it hard to use large proteins to treat disease.
“Delivery of therapeutic molecules is a major bottleneck for medicine.”
Feng Zhang
The inspiration: Bacteria have been around for billions of years, and they have evolved an amazing toolkit that scientists love to borrow from. (CRISPR, the gene editing tool, was originally discovered in bacteria.)
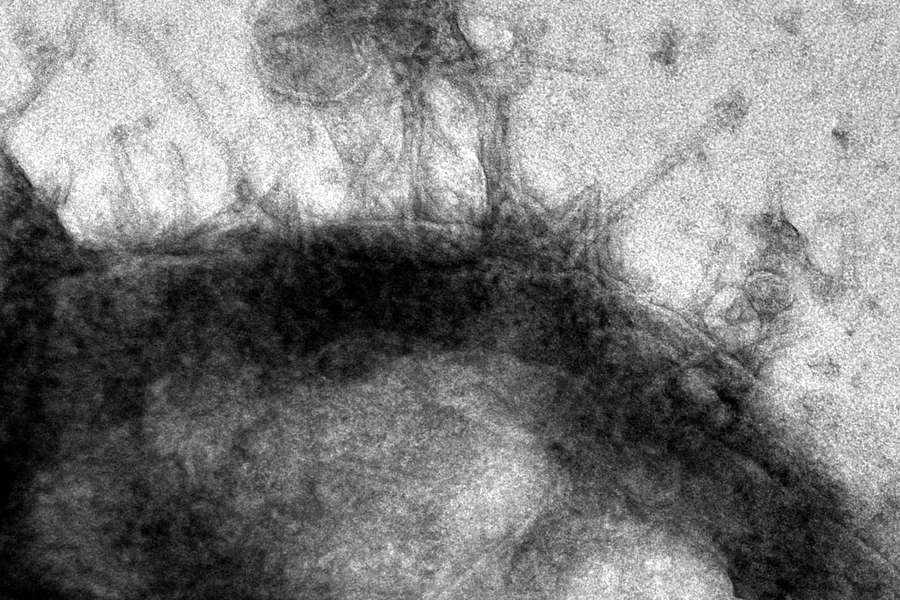
Some bacteria that live inside other creatures have a tiny mechanism for injecting proteins into other cells. Researchers at MIT have now found a way to use that tool, called an “extracellular contractile injection system” (eCIS), to insert proteins into human cells and even the brain cells of living mice.
“Delivery of therapeutic molecules is a major bottleneck for medicine, and we will need a deep bench of options to get these powerful new therapies into the right cells in the body,” said lead researcher Feng Zhang (also one of the early pioneers of CRISPR). “By learning from how nature transports proteins, we were able to develop a new platform that can help address this gap.”
How it works: The system is a kind of microscopic syringe, which can recognize the specific cells it’s supposed to inject.
It consists of a 100-nanometer tube, with tiny fibers on one end. When those fibers come into contact with the receptors on a target cell, they latch onto it — and then a flexible sheath on the outside of the tube contracts.
This drives a spike in the tube straight into the target cell so that it can deposit a protein payload.
“It doesn’t seem to kill cells that don’t contain the receptor that you’re targeting.”
Joseph Kreitz
For their study, the MIT team used the artificial intelligence tool AlphaFold to redesign the fibers on one type of bacterial eCIS — called the “the Photorhabdus virulence cassette” (PVC) — so that the nanosyringes could identify and bind to various human or mouse cells.
With some additional engineering, they figured out how to get the systems to include a protein payload of their choosing.
The results: To demonstrate potential medical uses for the nanosyringes, the researchers created examples capable of delivering various proteins, including ones that kill cancer cells, into human cells in the lab.
“It doesn’t seem to kill cells that don’t contain the receptor that you’re targeting,” researcher Joseph Kreitz told Genetic Engineering News.“So, you could imagine an application where you’re giving patients a PVC that targets a cancer epitope and then having the system go and deactivate cells that express that cancer epitope.”
They also injected loaded nanosyringes into the brains of live mice to deliver proteins into the rodents’ neurons. The system didn’t trigger a detectable immune response in the animals, either — that’s very important, because a severe immune reaction can be toxic.
Looking ahead: This is still very early research, but the MIT team sees tremendous potential for their nanosyringes as a delivery vehicle for proteins and maybe even other payloads — if we can figure out how to load them up with DNA and RNA, they could potentially be used to delivery gene therapies instead of immune system-triggering viral vectors.
“We’re just looking at the tip of the iceberg,” Zhang told GenEng. “It’s pretty trivial to re-engineer and is similar to producing any recombinant protein, but in terms of developing this into a biomedical tool, there’s still a lot of work to be done across the board, from discovery to manufacturing.”
We’d love to hear from you! If you have a comment about this article or if you have a tip for a future Freethink story, please email us at [email protected].






