After a brief interrogation by a bobby at the train station, Alastair Fraser-Urquhart walks out into an empty London. The streets are devoid of people, a city as vibrant as any in the world — a global capital — silenced. It is early January 2021, and the UK has entered full COVID lockdown. It is eerily quiet.
Except for the ambulances.
Fraser-Urquhart is heading to Imperial College London (ICL) — to prepare to deliberately inhale the very virus that everyone else in the country was shut indoors to try to avoid.

A Human Challenge
At the college, he is subject to a battery of tests. Height, weight, blood draws, a nasopharyngeal swab just to test his ability to stand them — to check his propensity for tears or a bloody nose: it goes deep, deep, then into the trash — and a machine for lung function that he has to breathe hard into, like really hard, as hard as he can. (The machine shames him, declaring “low effort” in a test to get a baseline for lung function.)
Fraser-Urquhart is one of a handful of people selected to participate in a “human challenge” trial for COVID-19 run by scientists at ICL, which intentionally exposes or “challenges” volunteers with the virus in order to study it.
The study, published in March 2022 in Nature Medicine, was the result of a long process — riven with scientific and ethical conflicts — kicked off by a controversial proposal in the Journal of Infectious Diseases at the beginning of the pandemic in 2020.
In that article, Rutgers bioethicist Nir Eyal, Harvard epidemiologist Marc Lipsitch, and Peter Smith, professor of tropical epidemiology at the London School of Hygiene and Tropical Medicine, proposed using human challenge trials to “accelerate the testing and potential rollout” of COVID-19 vaccines.
“I think there is a very important ethical debate that needs to be had,” Andrew Catchpole, the chief scientist at research firm hVIVO and an author on the ICL study, told me in April 2020. “Certainly, we are in unprecedented times, and in unprecedented times unprecedented measures are required.”
Rather than spending months recruiting tens of thousands of participants to get a vaccine or a placebo, and then waiting several months for a handful to be inevitably infected in the real world, you could work with a much smaller group, expose them to the virus in a lab, and see what works right away.
“I don’t know whether this idea is going to fly,” Smith told me previously. “But given the conversations I’ve had so far, I think it’s going to be taken very seriously.”
It tasted “like a school science lab.”
The proposal didn’t get far in the US, but the UK ran with it. Challenge studies were eventually run by ICL in early 2021 — to figure out the safety of such trials and study the infection’s early stages — and by Oxford soon thereafter, which ran a reinfection trial, challenging people who had previously recovered from the virus. The Oxford study has yet to be published.
Like any scientific tool, challenge trials have their strengths and weaknesses. By guaranteeing infection, they can accelerate the testing of vaccines and therapies, and comparing one to another quickly. They can standardize infection so researchers can better characterize what a pathogen is up to, or to test, say, how an infection spreads between people.
When the diseases are potentially deadly, they could also severely sicken, or even kill, their subjects. As a result, there are numerous ethical bars that need to be cleared, in addition to the scientific challenges of trial design and other surprisingly tricky problems, like growing the right virus to use.
Boiled all the way down, the debate comes down to this: can we get people sick with relatively little risk, and would what we learn from that outweigh the inherent risk?
Essentially, when it comes to the COVID-19 challenge trials, was the juice worth the squeeze?
The Challenge Experience
In March 2021, just before he’s about to get COVID on purpose, Fraser-Urquhart realized something: this is about the scariest thing he’s ever done in his life, up to that moment. (He now thinks his fear was unusual, based on other participants he’s spoken to.)
In the room where Fraser-Urquhart gets challenged, eight or nine people are standing around, wearing enclosed positive pressure hoods — some to handle samples, some to verify them, and one to count down the seconds, “as if it wasn’t horrifying enough,” he says.
He experienced a weird cognitive dissonance.
“Outside is devastation,” he says. “I went in, and it was like full-on lockdown … because of this virus. And then suddenly it’s literally there in the room.”

It felt like a physical presence, he says — odd, very odd.
He lay down on his back, hung his head off the edge of the table, and the researchers dropped a clear liquid containing SARS-CoV-2 into his nose.
“You can feel it running down, and it’s like fuck,” Fraser-Urquhart said — this is what everyone is desperately trying to avoid.
It tasted “like a school science lab.”
Opening the Black Box
In the room that day at the Royal Free Hospital was Christopher Chiu, an infectious diseases physician and immunologist at ICL, and lead researcher of the trial, the first human challenge trial for SARS-CoV-2.

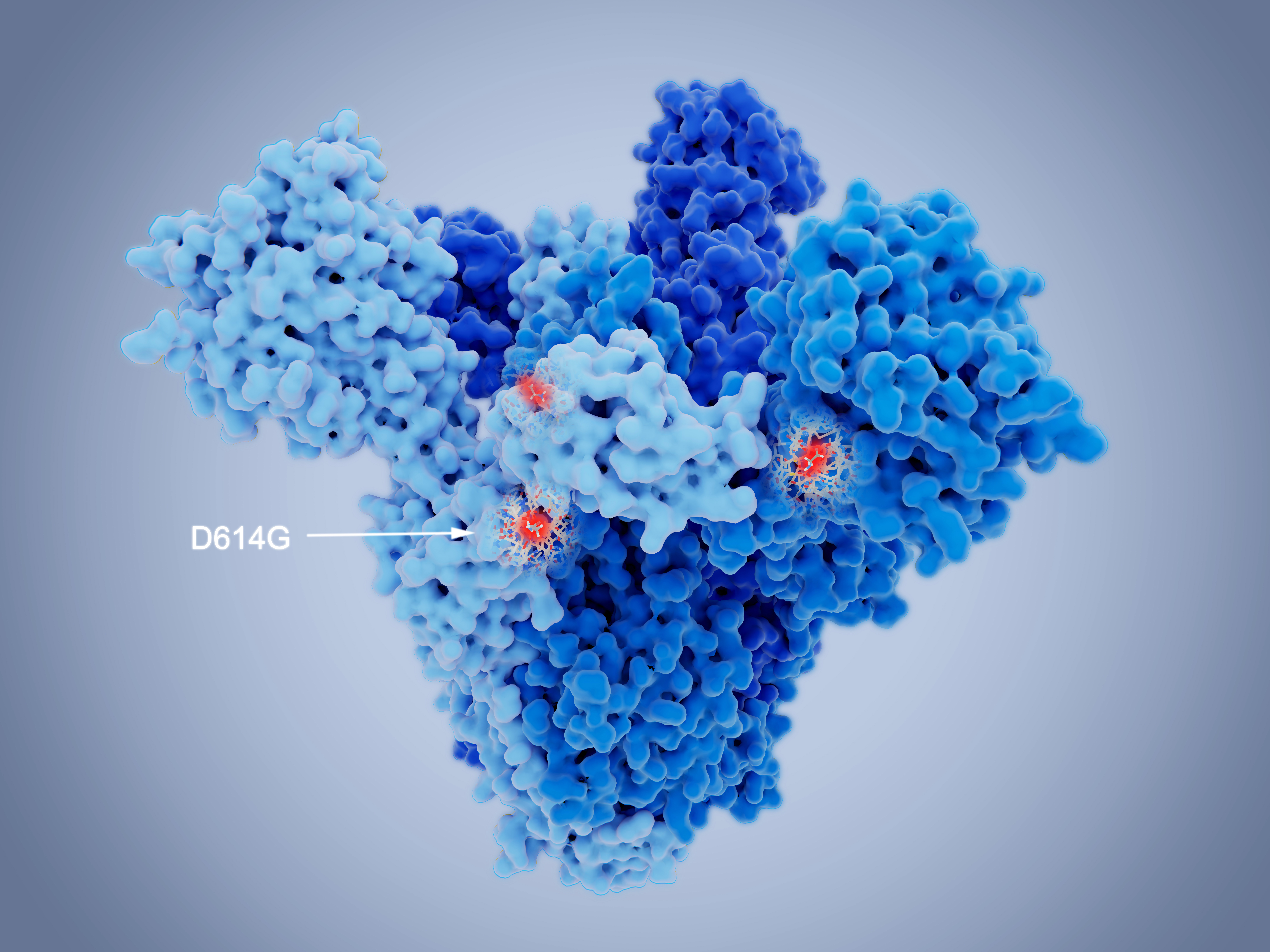
All of the study participants received the exact same virus (very similar to the original coronavirus strain, but with the D614G mutation), the exact same amount of virus, and in the exact same way — creating a perfectly standardized infection that is impossible to achieve in the real world.
Chiu wanted to leverage this unique situation to understand the very earliest period of infection.
“The things which we really can’t understand in natural infection are what happens right at the beginning of infection, between the time that you are exposed to the virus and the time that you start to develop symptoms,” Chiu says.
“That’s sort of a black box to us in most types of studies.”
This initial window is quite important. It could be the crucial time for the body’s immune system to rid the body of the virus, a window that’s poorly understood, since almost everyone who gets tested for COVID-19 already has symptoms. If we understood this phase better, we could likely make better vaccines and treatments, Chiu says.
To start, the team wanted to be able to better understand the course of infection, especially in mild cases — the people most likely to be the major drivers of transmission, Chiu says.
In addition, they tested how accurate lateral flow rapid antigen tests were at the beginning and end of infection, to help public health officials figure out when people could end self-isolation and how often people needed to re-test.
What We Learned
The study did yield insights into the early stages of infection: how much virus was necessary to be infected, how long did the virus need to incubate, when were people most likely to be contagious — insights difficult or near impossible to ascertain in the wild.
“What really surprised us first of all was how little virus was needed to cause infection,” Chiu says. Even the smallest dose of virus that could be reliably quantified was enough to cause infection in over half of challenged participants.
The second surprise was how rapidly the infection progressed.

Incubation periods for viruses can range from days to years. This coronavirus, in these patients, delivered in this way, fell on the “very short” end of the spectrum. Estimates up until that point, from studying hospitalized patients, pegged the incubation period at around five days, Chiu says.
But challenge participants began having detectable amounts of virus in their upper respiratory tract by day two, on average.
“That makes the likelihood of transmitting infection to other people before you develop symptoms quite a lot higher,” Chiu notes.
They also found high amounts of virus being “shed,” or released from the body, by talking, coughing, breathing, etc. While evidence for high shedding was found in other studies, whether or not symptoms correlated with shedding had been a point of contention — if being more symptomatic meant more infectious.
“What we actually found was that there was no correlation,” Chiu says. “Everybody shed huge amounts of virus,” even those with no symptoms at all.
The team has another paper, in preprint now, analyzing the environment of the study participants while they were in quarantine, where they found massive amounts of virus had been shed into their surroundings — a likely driver of SARS-CoV-2’s infectiousness.
The Push and the Questions
Fraser-Urquhart believes that HCTs are a valuable tool worth pursuing. In addition to pursuing his degree in biomedicine at University College London, he also works as a research assistant for 1Day Sooner, a nonprofit that advocates on behalf of aspiring participants in “high impact medical studies,” especially HCTs.
1Day Sooner was founded in March 2020 as a grassroots effort to represent volunteers willing to be infected to help make COVID-19 vaccines available, well, sooner.
The group has since branched into broader advocacy for trials, as well as an important voice for those who are too often left out of HCT decision making: the volunteers themselves.
Reorienting research around participants is a sea change; sources interviewed for this story believe it will continue to impact the field of infectious disease.
There’s a recent push by Malawian volunteers to do challenge studies to find new drugs and vaccines for TB, a disease that takes a huge toll throughout the developing world.
The new level of willing participants’ organization marks the difference, says Euzebiusz Jamrozik, a postdoctoral fellow at Oxford and the co-author of “Human Challenge Studies in Endemic Settings: Ethical and Regulatory Issues.”
Those voices have now appeared in journals like PLOS One, in a 1Day Sooner-sponsored paper, which found in a survey of volunteers that participants are not driven by money and don’t have a poor conception of their risks.
Challenge trials “show us that there are people who are truly happy to take on physical risks to advance human knowledge and health,” Fraser-Urquhart wrote for the Washington Post in June 2021.
“Denying us the opportunity to do so will only perpetuate suffering. Just as there will be more pandemics, there will be more volunteers. We have a lot to lose by ignoring them.”

But Did It Help?
Despite the participants’ unity in believing in the potential value of human challenge trials, there’s division about just how useful they have been, in fact, for SARS-CoV-2.
In their original proposal, Eyal, Lipsitch, and Smith proposed human challenge trials as a way to speed up vaccine development.
This didn’t happen.
Instead, spurred by billions in government funding, a variety of shots made their way into traditional phase 3 trials with record speed. By the time Fraser-Urquhart arrived in an eerily silent London, in January 2021, vaccines from Pfizer-Biontech, Moderna, and Oxford-AstraZeneca had already completed their trials and been approved by regulators.
Moreover, the virus that ICL was growing for its belated study was already a bit out of date, ceding the stage first to the Alpha variant — sweeping through the UK at the time — followed by Delta, then Omicron.
Three years after their proposal, the SARS-CoV-2 challenge trials have been “a damp squib,” argues Charles Weijer, a professor and bioethicist at Canada’s Western University.
Weijer opposed challenge studies for COVID-19 back in 2020, and still believes they’re the wrong tool for COVID-19 (and potentially other new viruses) because synthesizing the viral strains for challenge studies takes too long.
The time it would take for strain and challenge model development was not appreciated in the early days of the pandemic, Weijer believes, and that’s before the difficult process of creating your challenge virus.
An article in Nature Medicine, by pediatrician Kathryn Edwards of the Vanderbilt University School of Medicine and vaccinologist Kathleen M. Neuzil from the University of Maryland School of Medicine, supports that point. If HCTs were to be used to study each emerging variant of concern, the process of synthesizing the virus to the high standards necessary for clinical work “would be expensive and potentially slow,” they write, and this is already a problem with influenza challenge models.
But Chiu believes there are generalizable insights about COVID-19 to be gained even in out-competed strains.
“At the end of the day, these are all basically the same virus,” he says. “The majority of their biology and processes are going to be the same.”
For his part, Fraser-Urquhart feels the delay between proposal, implementation, and publication blunted the effectiveness of the ICL trial: “ethical hand-wringing” had managed to “minimize the benefits and maximize the risk” of the study.
“I think, as a volunteer, you could decide to be extremely pissed off about that,” he says.
The Future
Underpinning all human challenge trials is an inherent tension between two variables: the research benefits versus the safety of the participants.
Chiu, Weijer, Jamrozik, and every other researcher and bioethicist I’ve interviewed are always in agreement on this point: the safety and the full, willing, and informed consent of the subjects are paramount.
But pushing up against that is the reality that some of the most useful information a HCT could give you, at least during a pandemic, would come from doing them as fast as possible.
If a basic HCT model had been established at the outset of the pandemic — or before — it could have been a valuable tool for gauging test accuracy; could have revealed how little virus was needed for an infection and that shedding is high well before symptoms arrive; could have given quick, hard data on the efficacy of non-pharmaceutical measures, like masking; could have accelerated testing of new drugs and vaccines.
It could have also risked the lives of its volunteers using a virus that was yet to be very well understood — the inherent tension.
In the kind of public health future 1Day Sooner and other supporters of increased HCT use envision, there is something akin to a HCT platform, a basic model that can be plucked from the shelf and adjusted for the pathogen.
“I think we need to build capacity for this type of research,” Jamrozik, the Oxford postdoc, says, “so that it can be done before, during, and after a crisis.”
To him, it is the research before a crisis that may be the most important. He offers the UK’s “Common Cold Unit” as an example. This research unit ran human challenge trials on cold-causing pathogens from 1946 to 1985, providing important insights into the epidemiology, prevention, and treatment of the cold — and was the first to isolate a coronavirus.
A similarly supported approach to study SARS-CoV-2 and other respiratory viruses may help us to be prepared for the next pandemic.
“These are highly specialized studies that require expert staff and quarantine facilities,” Chiu says. “We have learned a lot about how to set up a model during an emergency so we will take that learning forward but rely on funding and institutional commitment to sustain the infrastructure.”
HCTs could still yet be a useful tool for studying SARS-CoV-2, in Jamrozik’s opinion. They could be especially useful in contrasting current variants and older variants that are no longer in the wild to help understand how they’re different.
For Weijer, who says he isn’t opposed to HCTs in general, laying the groundwork with interdisciplinary teams that can take into account the array of challenges — from pathogen development to participant safety to broader public health to ethical concerns — will be crucial to expanding HCT use while avoiding a potential nightmare scenario: a severe illness or death in an HCT, damaging to public trust.
If a healthy volunteer in an HCT for a vaccine dies, regardless of the true cause of death, it will likely be the vaccine that would be blamed, he believes.
How we fit human challenge trials into public health research is a question that must be confronted, because it is not hypothetical.
“The field is growing,” Chiu says. “There are an increasing amount of human challenge studies being done.”
HCTs have been used within the past few years for testing malaria antibody therapies and new vaccines for hookworm and RSV, among other things.
Balancing speed and safety, scientific and ethical challenges, and identifying the best use cases will all determine HCTs role in the future. The mobilization of volunteers like Fraser-Urquhart and advocates like 1Day Sooner will perhaps have a lasting impact on how those benefits and risks are weighed, and by whom.
But in the end, only one thing is certain: we will be challenged by another pandemic pathogen, purposefully or not. We can only do our best to be ready to combat it when we are.
We’d love to hear from you! If you have a comment about this article or if you have a tip for a future Freethink story, please email us at [email protected].